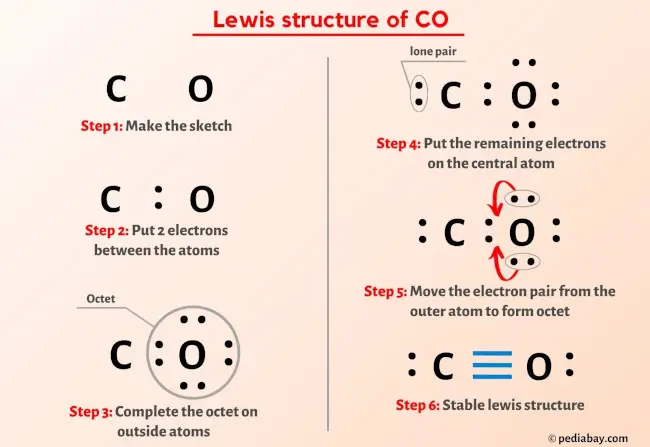
So you have seen the above image by now, right?
Let me explain the above image in short.
CO lewis structure has one Carbon atom (C) and one Oxygen atom (O) which contain a triple bond between them. There is 1 lone pair on both the Carbon atom (C) as well as Oxygen atom (O).
If you haven’t understood anything from the above image of CO (carbon monoxide) lewis structure, then just stick with me and you will get the detailed step by step explanation on drawing a lewis structure of CO.
So let’s move to the steps of drawing the lewis structure of CO.
Steps of drawing CO lewis structure
Step 1: Find the total valence electrons in CO molecule
In order to find the total valence electrons in CO (carbon monoxide) molecule, first of all you should know the valence electrons present in a single carbon atom as well as oxygen atom.
(Valence electrons are the electrons that are present in the outermost orbit of any atom.)
Here, I’ll tell you how you can easily find the valence electrons of carbon as well as oxygen using a periodic table.
Total valence electrons in CO molecule
→ Valence electrons given by carbon atom:
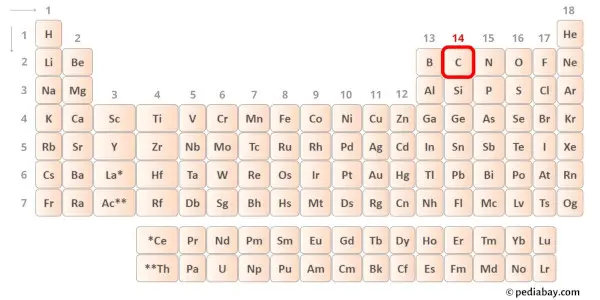
Carbon is group 14 element on the periodic table. [1] Hence the valence electrons present in carbon is 4.
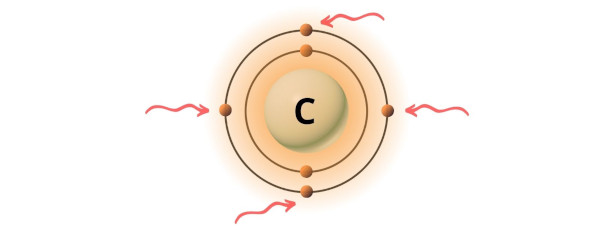
You can see the 4 valence electrons present in the carbon atom as shown in the above image.
→ Valence electrons given by oxygen atom:
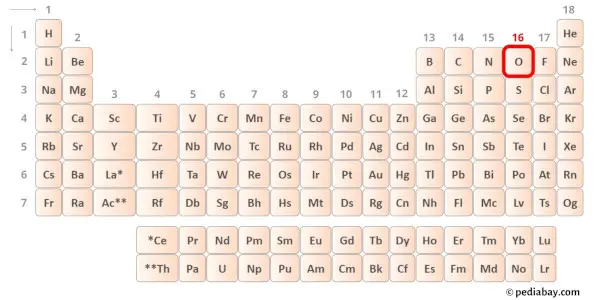
Oxygen is group 16 element on the periodic table. [2] Hence the valence electrons present in oxygen is 6.
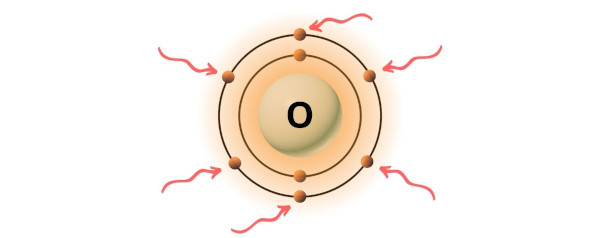
You can see the 6 valence electrons present in the oxygen atom as shown in the above image.
Hence,
Total valence electrons in CO molecule = valence electrons given by 1 carbon atom + valence electrons given by 1 oxygen atom = 4 + 6 = 10.
Step 2: Select the central atom
For selecting the center atom, you have to remember that the atom which is less electronegative remains at the center.
Now here the given molecule is CO (carbon monoxide). It has only two atoms, so you can select any of the atoms as a center atom.

Let’s assume the carbon atom as a central atom.
(You should assume the less electronegative atom as a center atom).
Step 3: Connect each atoms by putting an electron pair between them
Now in the CO molecule, you have to put the electron pairs between the carbon atom (C) and oxygen atom (O).

This indicates that the carbon (C) atom and oxygen (O) atom are chemically bonded with each other in a CO molecule.
Step 4: Make the outer atoms stable. Place the remaining valence electrons pair on the central atom.
Now in this step, you have to check the stability of the outer atom.
Here in the sketch of CO molecule, we have assumed the carbon atom as a center atom. So the oxygen is the outer atom.
Hence you have to make the oxygen atom stable.
You can see in the below image that the oxygen atom is forming an octet and hence it is stable.
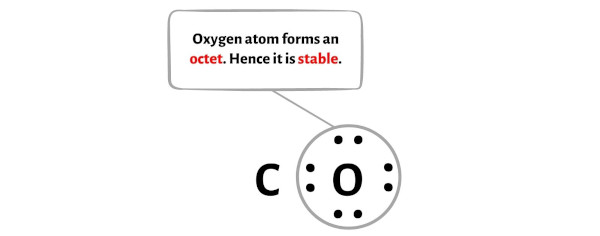
Also, in step 1 we have calculated the total number of valence electrons present in the CO molecule.
The CO molecule has a total 10 valence electrons and out of these, only 8 valence electrons are used in the above sketch.
So the number of electrons which are left = 10 – 8 = 2.
You have to put these 2 electrons on the carbon atom in the above sketch of CO molecule.

Now let’s proceed to the next step.
Step 5: Check the octet on the central atom. If it does not have octet, then shift the lone pair to form a double bond or triple bond.
In this step, you have to check whether the central carbon atom (C) is stable or not.
In order to check the stability of this carbon (C) atom, we have to check whether it is forming an octet or not.
Unfortunately, this carbon atom is not forming an octet here. Carbon has only 4 electrons and it is unstable.

Now to make this carbon atom stable, you have to shift the electron pair from the oxygen atom.
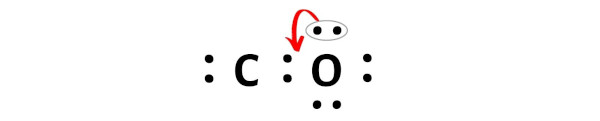
But after shifting one electron pair, the carbon atom is still not forming an octet as it has only 6 electrons.
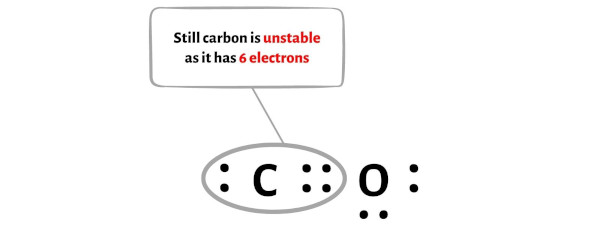
So again we have to shift one more electron pair from the oxygen atom.
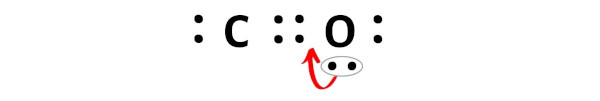
After shifting this electron pair, the carbon atom will get 2 more electrons and thus its total electrons will become 8.
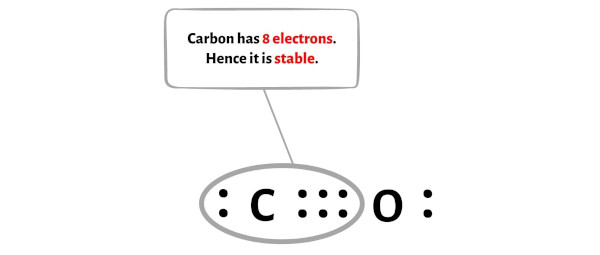
You can see from the above picture that the carbon atom as well as oxygen atom both are forming an octet.
And hence the above lewis dot structure of CO molecule is stable.
In the above lewis dot structure of CO, you can also represent each bonding electron pair (:) as a single bond (|). By doing so, you will get the following lewis structure of CO.
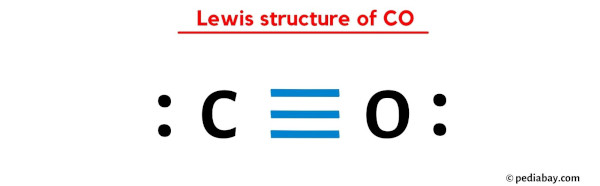
I hope you have completely understood all the above steps.
For more practice and better understanding, you can try other lewis structures listed below.
Try (or at least See) these lewis structures for better understanding:
| BF3 lewis structure | NO3- lewis structure |
| O3 lewis structure | C2H2 lewis structure |
| CH2O lewis structure | SO3 lewis structure |
Jay is an educator and has helped more than 100,000 students in their studies by providing simple and easy explanations on different science-related topics. He is a founder of Pediabay and is passionate about helping students through his easily digestible explanations.
Read more about our Editorial process.